Why Should We Concern the Temperature Effects on Solder Paste Flux
Why Should We Concern the Temperature Effects on Solder Paste Flux
As part of solder paste formulations, flux is an essential material for improved soldering effect. Flux can remove the oxide layer, change the soldering surface tension, and adjust the viscosity of the solder paste. Therefore, the choice of flux is very important for solder paste printability and dispensing. It is well known that temperature is an important factor affecting chemical reactions. Thus, temperature also influences flux activity.
Ambient Temperature Affects Flux Quality
In most cases, the temperature will speed up the rate of chemical reactions. If the temperature is not properly controlled during storage, the properties of the flux composition of the solder paste will change and affect the solderability. Therefore, solder paste generally needs to be stored in a refrigerator at 0-10 °C. In addition, the production workshop temperature needs to be controlled below 30 °C. Since the printing process requires a long stencil life, prolonged high temperatures will accelerate the volatilization of the solvent, causing the solder paste to dry out and viscosity reduction.
Reflow Soldering Temperature Effect
The active temperature range of the flux needs to cover the entire reflow process. The heating rate and dwell time during reflow can affect the performance of the solder paste flux. For lead-free solder paste, the preheating stage usually lasts tens to hundreds of seconds, and the heating rate is 1.5℃-3℃. In the preheating zone, an excessively fast heating rate will cause the flux solvent components to volatilize rapidly, and the pads will not be fully wetted, resulting in an uneven shape of the solder joints. The heating rate in the reflux zone is slightly different, usually controlled at about 2.5℃-3℃. After undergoing preheating zone and the soak zone, the solder paste temperature is high, and the flux components are constantly volatilizing. In order to better assist the wetting, a higher heating rate is required to ensure that the remaining flux can still play a wetting effect when the melting point of the solder paste is reached.
Effect of Solder Paste Flux Formulation Temperature
The formulation temperature at which the flux is formulated also affects the flux performance. For example, thixotropic agents are easily decomposed at high temperatures. If the formulation temperature is too high, the viscosity of flux will decrease because of thixotropic decomposition, resulting in a thermal slump. Kwon et al. (2019) produced solder paste with SAC305 solder powder and rosin flux and studied the effects of formulation temperature. Kwon et al. chose formulation temperatures of 100°C, 150°C, and 200°C. Under heating conditions of 150°C, the solder paste slump ratio increased with flux formulation temperature (Figure 1). It can be known that when the formulation temperature is higher, the slump resistance of the solder paste is worse. In addition, it can be found from Figure 2 that the solder paste with a high formulation temperature will cause a more significant solder paste bridging impact.
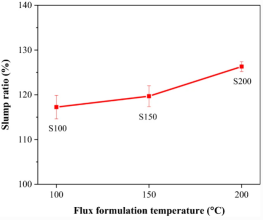
Figure 1.Relationship between Thermal Slump Ratio and Flux Formulation Temperature (Kwon et al, 2019).

Figure 2. Bridging condition after SAC305 solder paste curing, formulation temperature:(a)100℃; (b)150℃; (c)200℃ (Kwon et al, 2019).
Fitech is a world-leading provider of microelectronics and semiconductor packaging materials solutions. Fitech’s solder paste, epoxy solder paste, and solder powder have excellent wetting effects, narrow particle size distribution, and outstanding reliability after soldering. Welcome to Fitech’s official website for more information.
Reference
Kwon, S., Lee, H.J. & Yoo, S. (2019), “Effects of flux formulation temperature on printing and wetting properties of Sn–3.0Ag–0.5Cu solder”. Journal Materials Science: Materials in Electron 30, pp. 8493–8501.


















 Back to list
Back to list



