Introduction to PCB Creeping Corrosion_Shenzhen Fitech
Introduction to PCB Creeping Corrosion
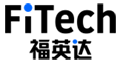
Creeping corrosion is a corrosion phenomenon on the surface of the circuit, which is generated by the reaction of corrosive gases such as sulphides or chlorides in the environment in contact with exposed copper. Creeping corrosion is characterised by solid corrosive material along the circuit and soldermask or encapsulation material surface migration growth, the formation of a structure similar to a spider's web, resulting in electrical shorts or impedance changes between adjacent pads or circuits, affecting the performance and reliability of electronic products. Creeping corrosion occurs from the exposed Cu surface. In environments containing sulphur substances, large amounts of black copper sulphides are generated on the Cu surface, which diffuse and accumulate on and around the Cu surface.
Figure 1. Creep corrosion on via pads (left) and magnified (right)
Copper oxides are insoluble in water, however copper sulphides and chlorides are soluble in water and have high surface mobility in response to concentration gradients. The products diffuse from areas of high concentration to areas of low concentration. Sulphides exhibit semiconducting properties and do not immediately cause short circuits, but as the concentration of sulphides increases, the hazards become apparent and eventually lead to short circuit failure.
In addition, the resistance value of the corrosion product will change dramatically with the temperature change, can be from 10MΩ down to 1Ω. Humidity (water film) will accelerate the creeping corrosion process: sulfide (such as sulphuric acid, sulphur dioxide) dissolved in water will generate a weak acid, the weak acid will cause the decomposition of the copper sulphide, forcing the clean Cu surface to be exposed, which continues to corrode. Obviously an increase in humidity accelerates this corrosion process. This corrosion occurs so quickly that some PCB veneers may even fail within a year.
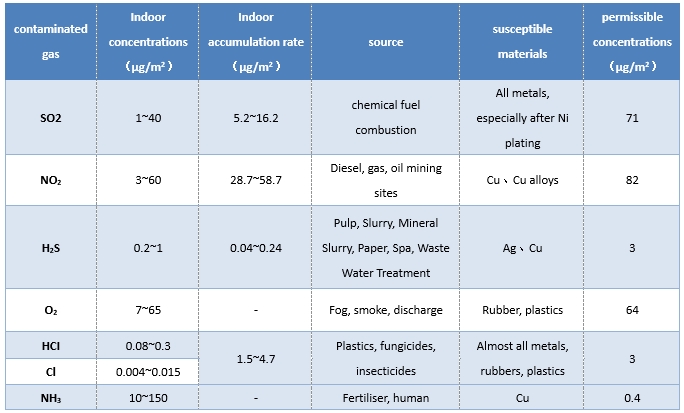
Atmospheric environment: The elements and gases that promote corrosion of electronic equipment as part of the atmospheric environment are listed as SO2, NO2, H2S, O2, HCI, Cl2, NH3, etc., and the indoor concentrations of corrosive gas constituents, the rate of accumulation, the source of occurrence, the impacts and susceptibility of the materials and permissible concentrations are shown in the table. As soon as the above gases are dissolved in water, corrosive acids and salts are easily formed.
Table 1. Indoor concentrations, accumulation rates, sources, susceptible materials and permissible concentrations of corrosive gas components.
Humidity: According to the dissolution/diffusion/deposition mechanism of creeping corrosion, an increase in humidity accelerates the occurrence of sulphide corrosion. According to Ping Zhao et al, the rate of creeping corrosion has an exponential relationship with humidity.Carig Hillman et al showed in mixed gas experiments that the corrosion rate increases sharply and parabolically with the rise of relative humidity. Cu, for example, when the humidity increases from 60% RH to 80%, the corrosion rate of the latter for the former 3.6 times.
Soldering process: Soldering process is also an important influencing factor. The thermal shock of reflow soldering may lead to localised tiny stripping of green oil or damage to certain surface treatments (such as OSP), increasing the risk of copper exposure to electronic products and thus increasing the likelihood of creeping corrosion.
PCB substrate and plating material: PCB substrate and plating material selection also has a significant impact on creepage corrosion. Studies have shown that in the Au / Pd / SnPb three plating structure, brass crawling corrosion resistance is the best, CuNi is the worst; SnPb surface treatment is the least susceptible to corrosion, and Au, Pd corrosion products on the surface of the longest crawling distance.Alcatel-Lucent's research found that the corrosion resistance of various surface treatments ranked ImSn ~ HASL>> ENIG>>OSP>>ImAg. Chemical silver itself does not cause creeping corrosion, but the probability of occurrence is higher in its surface treatment, because chemical silver PCB exposed copper or surface microporosity is more serious, making bare copper more susceptible to corrosion.
The use of three-proof coating, that is, the surface of the circuit is coated with a protective film to isolate the external corrosive gases;
Design and process to reduce the probability of exposed copper PCB and components, select the surface treatment process with strong corrosion resistance, such as lead-free hot air levelling, tin dipping, etc., to avoid the use of chemical silver, organic solder protection, etc.;
The assembly process should minimise thermal shock and contamination ion residues, choose suitable fluxes and carry out effective cleaning;
The whole machine design to strengthen the control of temperature and humidity, to avoid the formation of water film and the dissolution of corrosive substances;
The location of the machine room should avoid obvious sulphur pollution, such as industrial areas, volcanic areas and swamps.

















 Back to list
Back to list



